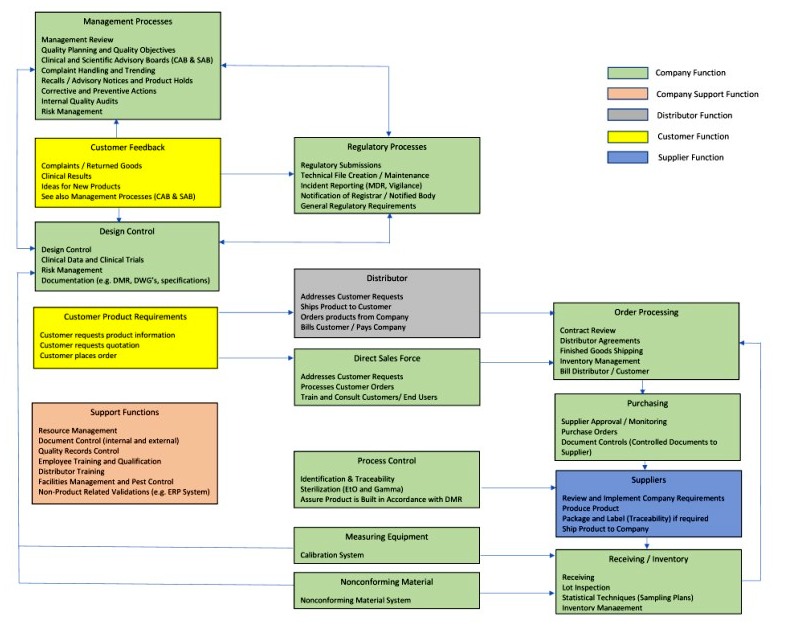
Iso 13485 quality manual

ISO 13485:2016 can also be used by suppliers or external parties that provide product, including quality management system-related services to such organizations. Such organizations can be involved in one or more stages of the life-cycle, including design and development, production, storage and distribution, installation, or servicing of a medical device and design and development or provision of associated activities (e.g. ISO 13485:2016 specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements. Objective evidence to support your personnel through the design,ĭevelopment, manufacturing, and support of medical devices.ISO 13485:2016 Medical devices - Quality management systems - Requirements for regulatory purposes Documentation is key for objectiveĮvidence. By now, you could get to the conclusion that documentation should beĪbout defining processes and maintaining the required records to demonstrate Standard and describes how the medical device company addresses the clauses.įew burdens and obstacles regarding documentation and it doesn’t have to be It breaks down the to various sections of ISO 13485:2016

#ISO 13485 QUALITY MANUAL MANUAL#
Organizations for satisfying this quality manual needs is creating a long-lasting Described interactions between the processes of the quality management system.